The global market for Self-Contained Biological Indicators (SCBI) is witnessing unprecedented growth, driven by stringent healthcare regulations and the rising volume of surgical procedures worldwide. As the "gold standard" for sterilization validation, SCBIs are essential for ensuring the total destruction of microbial life, including highly resistant spores like Geobacillus stearothermophilus.
The industry is shifting from traditional 48-hour incubation periods to Rapid Readout technology. Modern SCBIs can now provide definitive results in as little as 1 hour or 3 hours using fluorescence-based detection. This evolution allows hospitals and dental clinics to release sterilized loads faster, significantly improving operational efficiency.
Global procurement is heavily influenced by ISO 11138 standards. Manufacturers must demonstrate extreme precision in D-value and Z-value calculations. Our factory adheres to these international benchmarks, ensuring that every SCBI batch provides reliable, repeatable results for Steam, EO, and Plasma sterilization processes.
Guangdong Onyx Medical Co., Ltd. is a leading national high-tech enterprise specializing in the research, development, production, and supply of advanced materials for the personal care, medical, and electronics industries. Since its establishment, the company has adhered to a core philosophy of innovation and quality, dedicated to providing high-quality products and solutions to customers worldwide.
Located in Guangdong, China, the company operates a modern manufacturing facility covering over 60,000 square meters, equipped with three Class 100,000 cleanrooms and more than 30 PLC-controlled automated production lines. This state-of-the-art infrastructure ensures that every production process meets the highest levels of precision and safety, adhering to international standards.
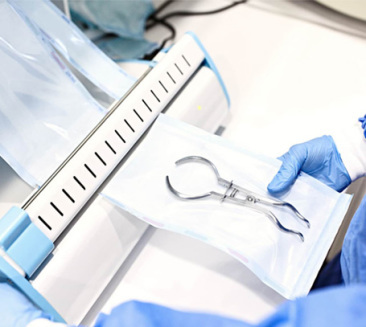


Central Sterile Supply Departments (CSSD) are the primary users of SCBIs. They require high-volume, reliable indicators for daily autoclave cycles to ensure patient safety and prevent HAIs (Healthcare-Associated Infections).
Smaller clinics increasingly adopt SCBIs due to their "all-in-one" design, which eliminates the need for a microbiology lab. Our 25PCS packs are ideal for localized, small-scale sterilization monitoring.
In drug manufacturing, sterilization validation is a critical regulatory hurdle. Our BIER (Biological Indicator Evaluation Resistometer) and high-precision SCBIs provide the data integrity required for pharma audits.
Guangdong Onyx Medical operates under the guiding principles of "Technological Innovation, Quality First," striving to establish a strong brand presence in the global market. As a China-based factory, we offer unique advantages:







