The Czech Republic has established itself as a central hub for medical excellence in Central Europe. With world-class facilities like the Motol University Hospital in Prague and the St. Anne's University Hospital in Brno, the demand for high-performance sterilization materials has never been higher. As a leading supplier, we understand that the Czech healthcare sector operates under the strict guidelines of the European Medical Device Regulation (MDR) and EN ISO 11607 standards.
Our SMMMS (Spunbond-Meltblown-Meltblown-Meltblown-Spunbond) non-woven wraps are engineered to meet these rigorous requirements, providing a five-layer microbial barrier that ensures the safety of surgical instruments from the autoclave to the operating theater.
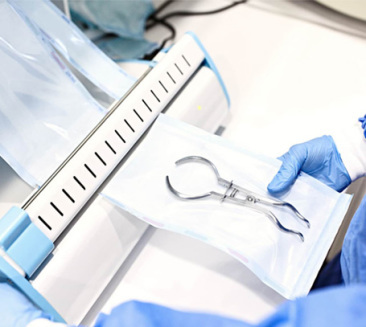
Understanding the layers of protection for modern Czech surgical environments.
SMMMS Structure for Maximum Filtration
Bacterial Filtration Efficiency (BFE)
Polypropylene Virgin Material
Certified Quality Management
The "M" layers (Meltblown) act as the core filter, trapping microorganisms, while the "S" layers (Spunbond) provide the structural integrity and durability required for heavy surgical trays. In the Czech Republic's industrial sterilization centers, where efficiency and safety are paramount, our SMMMS wraps reduce the risk of "wet packs" due to their excellent breathability and moisture vapor transmission rates.

In major Czech cities, CSSDs are moving towards high-volume automated sterilization. Our SMMMS wraps are designed to withstand the mechanical stresses of automated handling systems used in modern Czech clinics.
Beyond large hospitals, the growing private dental sector in Prague and Brno requires reliable, smaller-format sterilization wraps. Our 45g and 50g variants are perfect for delicate dental kits, ensuring they remain sterile for up to 180 days when stored correctly.
The Czech Republic's thriving biotech corridor (especially around Vestec) utilizes our non-woven materials for cleanroom applications and equipment protection, where lint-free properties are critical.
Guangdong Onyx Medical Co., Ltd. is a leading national high-tech enterprise specializing in the research, development, production, and supply of advanced materials for the personal care, medical, and electronics industries. Since its establishment, the company has adhered to a core philosophy of innovation and quality, dedicated to providing high-quality products and solutions to customers worldwide. Located in Guangdong, China, the company operates a modern manufacturing facility covering over 60,000 square meters, equipped with three Class 100,000 cleanrooms and more than 30 PLC-controlled automated production lines.

Guangdong Onyx Medical operates under the guiding principles of "Technological Innovation, Quality First," striving to establish a strong brand presence in the global market. The company places a strong emphasis on quality control while ensuring that its production processes are sustainable. With a commitment to environmental protection, resource utilization, and social responsibility, Onyx Medical has gained a reputation as a trusted partner in the healthcare, personal care, and electronics industries.
The Czech Ministry of Health is increasingly focusing on "Green Hospitals." This shift is driving the demand for recyclable medical consumables. Our SMMMS wraps are made from 100% Polypropylene, which is fully recyclable in the Czech Republic's advanced waste management systems. By choosing Onyx Medical, Czech providers can balance patient safety with environmental goals.
Furthermore, the integration of AI and Digital Tracking in Czech CSSDs means that sterilization wraps must now often accommodate RFID tags or advanced barcode labels. Our material surface is optimized for high-adhesion sterilization tapes and labels, ensuring no data loss during the high-pressure steam cycles common in Czech autoclaves.
Ready to upgrade your Czech medical facility's sterilization protocols?
Our logistics network ensures timely delivery to Prague, Brno, and Ostrava, minimizing supply chain disruptions. We provide full technical documentation in English and can assist with local regulatory compliance. Our Class 100,000 cleanrooms ensure that every sheet of SMMMS wrap is free from contaminants before it even reaches your facility.
 Product line
Product line Product line
Product line Product line
Product line Product line
Product line