In the current global health climate, Australia’s healthcare and life sciences sectors are experiencing a period of rigorous modernization. From the bustling medical hubs in Melbourne’s Parkville precinct to the expansive hospital networks across New South Wales and Queensland, the demand for precision sterilization monitoring is at an all-time high. Self-Contained Biological Indicators (SCBI) have become the "gold standard" for verifying the efficacy of sterilization processes, ensuring patient safety and regulatory compliance.
Australia’s stringent regulatory environment, governed by the Therapeutic Goods Administration (TGA), mandates that medical and dental facilities adhere to AS/NZS 4187 and AS/NZS 4815 standards. These standards emphasize the need for biological monitoring to validate that every autoclave cycle successfully eliminates highly resistant microbial spores. Consequently, Australian manufacturers and distributors are increasingly seeking SCBIs that offer not only high sensitivity but also rapid readout capabilities to minimize equipment downtime.
TGA Compliance Readiness
Rapid Results Tech
sqm Production Base
The Australian sterilization industry is pivoting toward Smart Monitoring. The integration of SCBI results with hospital information systems (HIS) via automated readers is the next frontier. This reduces human error in recording results and provides a digital audit trail for accreditation purposes.
Sustainability is another major trend. Manufacturers are now looking at reducing the plastic footprint of SCBI vials without compromising the integrity of the Geobacillus stearothermophilus or Bacillus atrophaeus spore carriers. As Australia moves toward "Green Hospital" initiatives, eco-friendly medical consumables are becoming a significant competitive advantage.
Guangdong Onyx Medical Co., Ltd., as a global leader, recognizes these trends. By combining 100,000-class cleanroom manufacturing with PLC-controlled automation, we provide the Australian market with products that meet the highest international benchmarks of safety and reliability.
Guangdong Onyx Medical Co., Ltd. is a leading national high-tech enterprise specializing in the research, development, production, and supply of advanced materials for the personal care, medical, and electronics industries. Since its establishment, the company has adhered to a core philosophy of innovation and quality, dedicated to providing high-quality products and solutions to customers worldwide. Located in Guangdong, China, the company operates a modern manufacturing facility covering over 60,000 square meters, equipped with three Class 100,000 cleanrooms and more than 30 PLC-controlled automated production lines. This state-of-the-art infrastructure ensures that every production process meets the highest levels of precision and safety, adhering to international standards.
Guangdong Onyx Medical operates under the guiding principles of "Technological Innovation, Quality First," striving to establish a strong brand presence in the global market. The company places a strong emphasis on quality control while ensuring that its production processes are sustainable. With a commitment to environmental protection, resource utilization, and social responsibility, Onyx Medical has gained a reputation as a trusted partner in the healthcare, personal care, and electronics industries.

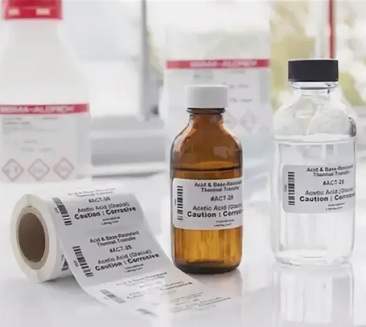









To the uninitiated, an SCBI might look like a simple vial, but it is a feat of biological engineering. Inside every Onyx Medical SCBI is a standardized population of bacterial spores—typically Geobacillus stearothermophilus for steam and plasma sterilization, and Bacillus atrophaeus for ethylene oxide. These spores are chosen for their extreme resistance to the specific sterilization agent being tested.
1. Placement: The SCBI is placed in the most challenging area of the autoclave load, often inside a Process Challenge Device (PCD).
2. Exposure: The sterilization cycle runs. The steam or gas must penetrate the vial’s filter to reach the spores.
3. Activation: After the cycle, the user "crushes" the vial to release the growth media into the spore chamber.
4. Incubation: The vial is placed in an incubator. If the spores were not killed, they germinate, changing the media color (usually from purple to yellow) or producing a fluorescent signal in rapid-readout versions.
For Australian providers, the Reliability of these indicators is non-negotiable. A "false negative" could lead to unsterile instruments being used on a patient, while a "false positive" leads to expensive and unnecessary re-sterilization of equipment. Onyx Medical’s SCBIs are manufactured under strict ISO 13485 quality management systems, ensuring that D-values and population counts are consistent every single time.
While based in Guangdong, our reach is global. We understand that the Australian market requires more than just a product; it requires a partnership. We provide full documentation support for TGA inclusion, including certificates of analysis (CoA) for every batch. Our logistical network is optimized to ensure that temperature-sensitive indicators are shipped via climate-controlled channels to maintain the viability of the spores during their journey across the Pacific.
Whether you are a dental supply distributor in Melbourne, a hospital procurement officer in Sydney, or a medical device manufacturer in Adelaide, Guangdong Onyx Medical Co., Ltd. offers the technological edge and manufacturing scale to support your sterilization monitoring needs.