High-precision monitoring tools optimized for the South American clinical environment.
Uruguay has emerged as a sophisticated hub for healthcare and pharmaceutical excellence in the Southern Cone. With a strong regulatory framework governed by the Ministerio de Salud Pública (MSP), the demand for high-quality Self-Contained Biological Indicators (SCBI) has reached an all-time high. Local hospitals in Montevideo, Salto, and Punta del Este are increasingly transitioning from traditional spore strips to advanced SCBI technology to ensure patient safety and operational efficiency.
The industrial landscape in Uruguay is shifting toward Industry 4.0, where sterilization processes are no longer just about killing bacteria, but about data integrity and rapid verification. Our SCBI products are designed to meet the rigorous ISO 11138 standards, providing Uruguayan laboratories and CSSDs (Central Sterile Supply Departments) with the reliable evidence they need for audit compliance.
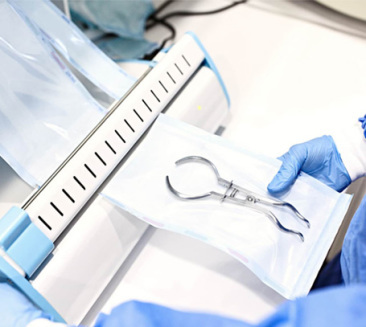
Advanced Sterilization Packaging Series for Uruguay
Guangdong Onyx Medical Co., Ltd. is a leading national high-tech enterprise specializing in the research, development, production, and supply of advanced materials for the personal care, medical, and electronics industries. Since its establishment, the company has adhered to a core philosophy of innovation and quality, dedicated to providing high-quality products and solutions to customers worldwide. Located in Guangdong, China, the company operates a modern manufacturing facility covering over 60,000 square meters, equipped with three Class 100,000 cleanrooms and more than 30 PLC-controlled automated production lines. This state-of-the-art infrastructure ensures that every production process meets the highest levels of precision and safety, adhering to international standards.
Guangdong Onyx Medical is structured into multiple specialized divisions, each committed to addressing the unique needs of its respective industries:
Guangdong Onyx Medical operates under the guiding principles of "Technological Innovation, Quality First," striving to establish a strong brand presence in the global market. The company places a strong emphasis on quality control while ensuring that its production processes are sustainable. With a commitment to environmental protection, resource utilization, and social responsibility, Onyx Medical has gained a reputation as a trusted partner in the healthcare, personal care, and electronics industries. The company’s advanced research and development capabilities, coupled with its efficient production systems, continue to drive it forward as a key player in these sectors. Moving forward, Onyx Medical is dedicated to pushing the boundaries of material science, providing innovative and efficient solutions for its customers worldwide.
Tailored solutions for the unique industrial landscape of Uruguay.
Uruguay's unique "Mutualista" system demands high-volume, cost-effective sterilization monitoring. Our 48-hour and 3-hour SCBIs are ideal for the rapid turnover of surgical instruments in Montevideo's busiest clinics.
With a growing export sector, Uruguayan pharma companies must adhere to GMP and international standards. Our Geobacillus stearothermophilus indicators ensure that every autoclave cycle meets the highest global safety benchmarks.
From Punta del Este to Colonia, dental practices are adopting SCBIs for on-site autoclave verification, providing patients with visual proof of sterile safety through our integrated chemical indicators.
In the Uruguayan medical sector, time is a critical factor. Traditional biological indicators requiring 48 to 72 hours of incubation are being replaced by Rapid Readout SCBIs (1h, 3h, and 4h). This trend allows Uruguayan hospitals to release sterilized loads faster, reducing the inventory costs of surgical instrument sets.
Furthermore, the integration of Auto-Readers with digital logging is becoming the standard. This eliminates human error in color-change interpretation, providing an indisputable digital record for the Uruguayan health authorities (MSP). Our technology bridges the gap between traditional microbiology and modern digital health.

Our Precision Automated Production Line
Explore our full range of sterilization monitoring systems for diverse applications.
Experience and expertise driving the global supply of sterilization materials.

Automated Biological Cultivation

Quality Control Testing

Packaging & Logistical Hub
Precise populations of G. stearothermophilus (10^5 or 10^6).
Rapid results in as little as 1 hour for urgent surgical loads.
Fully compliant with Uruguayan import regulations and MSP standards.
Pairs seamlessly with automated incubation and reading systems.