In the modern industrial landscape, Package Integrity Testing Equipment has transitioned from a niche quality control step to a non-negotiable regulatory requirement. As global healthcare standards like ISO 11607 and FDA Title 21 CFR Part 11 become increasingly stringent, the demand for precision instruments that can verify the microbial barrier of medical packaging has skyrocketed.
Globally, the market is driven by the rise in chronic diseases, the expansion of surgical procedures, and a heightened focus on infection control following the global pandemic. Whether it is a hospital's Central Sterile Supply Department (CSSD) or a high-volume medical device manufacturer, ensuring that a package remains sterile until the point of use is a matter of life and death. This "Zero-Failure" mandate has fueled the growth of specialized factories and exporters capable of delivering high-accuracy testing solutions.
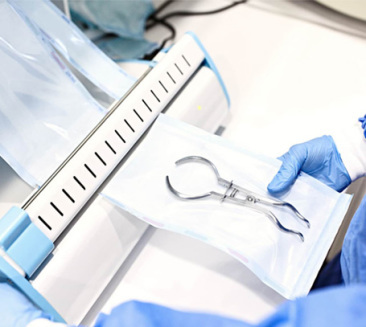
A Premier National High-Tech Enterprise

Guangdong Onyx Medical Co., Ltd. stands at the forefront of the advanced materials and medical testing industry. Headquartered in Guangdong, China, we operate a massive 60,000 square meter state-of-the-art manufacturing facility. Our infrastructure includes three Class 100,000 cleanrooms and over 30 PLC-controlled automated production lines, ensuring that every product—from a simple indicator strip to a complex plasma sterilizer—meets the highest international standards of precision and safety.
Since our inception, we have been dedicated to research, development, and the global supply of materials for healthcare, personal care, and electronics. Our philosophy, "Technological Innovation, Quality First," drives us to exceed the expectations of our partners across North America, Europe, and Asia-Pacific.
The industry is moving toward fully automated testing cycles. Our PLC-controlled equipment allows for real-time data logging and remote monitoring, reducing human error and enhancing traceability in sterile processing environments.
Sustainability is no longer optional. There is a massive trend toward biodegradable sterilization pouches and chemical-free indicators. Onyx Medical is investing heavily in "Green Biomaterials" to meet this demand.
Hospitals can no longer afford to wait 24-48 hours for biological indicator results. Our rapid reader incubators provide definitive results in a fraction of the time, accelerating the turnover of surgical instruments.
Healthcare & Medical Materials: Development of advanced biomaterials and sensors used in diagnostic equipment and vital signs monitoring. We focus on enhancing patient outcomes through material science.
Medical Sensors & Control Consumables: Manufacturing high-precision sensors that integrated into critical medical devices, ensuring reliability and safety in health monitoring systems.
New Biomaterials: Pioneering research in biocompatible materials for regenerative medicine and innovative drug delivery systems.
Diaper & Hygiene Materials: Providing the global market with high-quality, moisture-absorbent materials for infant and adult hygiene products, prioritizing comfort and safety.
New Electronics Materials: Engineering next-generation materials for consumer electronics, focusing on performance, durability, and thermal management.
As a leading Chinese manufacturer, Guangdong Onyx Medical offers a unique combination of scale, speed, and sophistication. The "Made in China 2025" initiative has transformed the medical manufacturing sector, enabling us to offer:
Our commitment to Environmental Protection and Social Responsibility ensures that your procurement process supports ethical and sustainable manufacturing practices.

In the Central Sterile Supply Department, our Bowie-Dick Test Packs and Steam Indicators are used daily to validate that every autoclave cycle successfully reaches the required parameters to kill all microbial life.
Small-scale sterilization is critical. Our Self-Sealing Sterilization Pouches and Gusseted Reels provide dentists with a reliable way to maintain instrument sterility from the autoclave to the patient's mouth.
During drug development, environmental monitoring is key. Our Biological Cooling Incubators allow researchers to cultivate and monitor biological indicators to ensure lab equipment remains uncontaminated.

Integrity testing is not just about having a machine; it is about understanding the science of sterilization. Our team consists of material scientists and medical engineers who understand the nuances of Hydrogen Peroxide Plasma Sterilization versus Ethylene Oxide (EO) methods.
Every batch of Class 4, 5, and 6 Indicator Strips undergoes rigorous internal testing against calibrated master cycles. This dedication to expertise and reliability is why we are trusted by Top 10 medical distributors worldwide. When you source from Onyx Medical, you are sourcing peace of mind.
Whether you are looking for a bulk supply of chemical indicators or high-end plasma sterilization equipment, Guangdong Onyx Medical is your reliable partner. We combine technical innovation with massive production capacity to serve the world's most demanding medical and industrial markets.
Optimized logistics from Guangdong port to your warehouse.
Rigorous Class 100,000 cleanroom manufacturing.
Custom packaging and branding solutions for your local market.







